-
Physical Classification Of Matter
Gaseous State
The matter in gaseous state has neither definite volume nor shape.
Examples : Air, oxygen, nitrogen, hydrogen, ammonia, carbon dioxide, compressed natural gas (CNG) etc.*Properties of Gases
(a) Gases neither have a definite shape nor a definite volume : Gases do not have a definite shape, but they acquire the shape of the vessel in which they are placed.
Similarly, gases do not have a definite volume, but attain the volume of container to which they are transferred.(b) Gases have maximum fluidity and least rigidity : The gases have high fluidity (property to flow) and least rigidity (tendency to maintain shape) due to large inter particle space and weak inter particle forces of attraction in them.
(c) Gases are highly compressible : The gases are highly compressible due to large inter particle spaces in them. Due to high compressibility, large volume of a gas can be compressed into a small cylinder and transported easily. e.g. L.P.G. gas & O2 supplied to hospitals in cylinders is compressed gas. Similarly these days, compressed natural gas (CNG) is used as a fuel in vehicles.
Gases are highly compressible while liquids are almost incompressible, while solids are completely incompressible.
-
Shape, Volume, Fluidity And Compressibility Of Gases
Gaseous State
The matter in gaseous state has neither definite volume nor shape.
Examples : Air, oxygen, nitrogen, hydrogen, ammonia, carbon dioxide, compressed natural gas (CNG) etc.*Properties of Gases
(a) Gases neither have a definite shape nor a definite volume : Gases do not have a definite shape, but they acquire the shape of the vessel in which they are placed.
Similarly, gases do not have a definite volume, but attain the volume of container to which they are transferred.(b) Gases have maximum fluidity and least rigidity : The gases have high fluidity (property to flow) and least rigidity (tendency to maintain shape) due to large inter particle space and weak inter particle forces of attraction in them.
(c) Gases are highly compressible : The gases are highly compressible due to large inter particle spaces in them. Due to high compressibility, large volume of a gas can be compressed into a small cylinder and transported easily. e.g. L.P.G. gas & O2 supplied to hospitals in cylinders is compressed gas. Similarly these days, compressed natural gas (CNG) is used as a fuel in vehicles.
Gases are highly compressible while liquids are almost incompressible, while solids are completely incompressible.
-
Experiment Illustrating Compressibility
Experiment to illustrate comparison between solids, liquid and gases in terms of compressibility.
or
Experiment to study the compressibility of solids, liquids & gases :
Take three syringes (about 100ml) and close their nozzles by rubber corks. Now remove the pistons from all syringes. Fill some water (liquid) in second syringe and chalk pieces (solid) in the third & leaving first syringe untouched. Now insert pistons back into syringes.
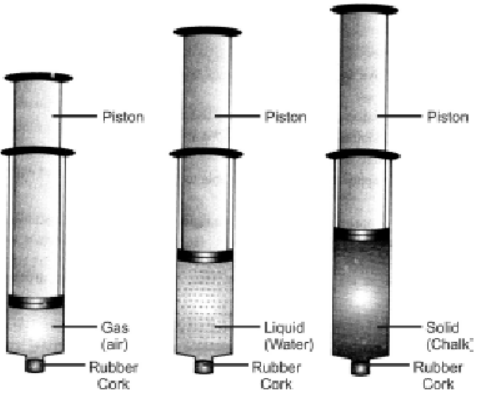
Observation and explanation: The piston of first syringe (left untouched) which contained air (gas) was easily pushed in. The piston of the second syringe which contained water (liquid) was pushed in only a little, while the piston of the third syringe which contained chalk pieces could not be pushed in at all. Thus, air is easily compressible, water is almost incompressible, while chalk pieces are completely incompressible.
Conclusion: The spaces between particles of gases are maximum, intermediate in liquids and minimum in solids. Thus, gases are highly compressible, liquids are almost incompressible, while solids are completely incompressible. -
Density, Kinetic Energy And Pressure Exerted by Gases
(d) Gases have low density : Gases have low density as compared to solids and liquids due to large inter molecular spaces in them. i.e. mass per unit volume of a gas is small and hence gases have low density.
(e) The Kinetic energy of particles in the gaseous state is quite high :- Due to large inter particle distances and weak forces of attraction, the particles of a gas can move freely & thus have large rotational, translational and vibrational motion and due to large translational motion, their kinetic energy is quite high which can be further increased by increasing the temperature of gas.
(f) Gases exert pressure : Due to larger inter particle distances and weaker inter particle forces of attractions, particles of a gas are moving continuously in different directions with different velocities. Due to this random motion, the particles of gas collide with each other and also with the walls of the containing vessel. Due to these collisions, the particles of the gas exert a force on the walls of the container. This force per unit area exerted by the particles of the gas on the walls of containing vessel is called the pressure of the gas.
Random motion means motion in different directions with different velocities. The random motion of particles of a gas is due to larger inter particle distances and weaker inter particle forces of attraction between them, unlike liquids & solids
The motion and inter particle distances in solids, liquids & gases are as shown in fig.
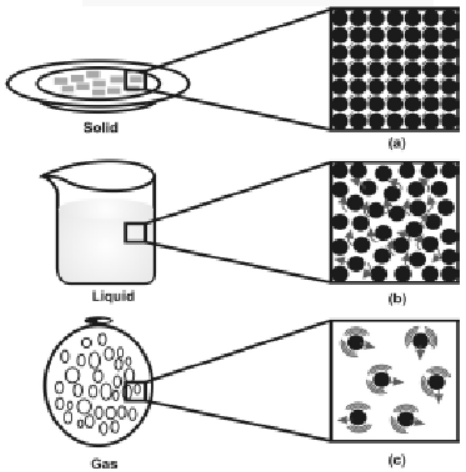
a, b and c show the magnified schematic pictures of the three states of matter. The motion of the particles can be seen and compared in the three states of matter.
(g) Gases diffuse very rapidly : Due to random motion, the particles of one gas readily move into spaces between the particles of the other gas. (called diffusion)
Thus, gases diffuse very rapidly, rate of diffusion increases with increase in temperature.
The most familiar example of diffusion of gases is found in our homes, e.g. we come to know what is cooked in the kitchen without even entering there, by the smell due to rapid diffusion. (i.e. rapid intermixing of particles of aroma with particles of air). Since rate of diffusion becomes faster at high temperature the smell of hot cooked food travels faster than that of the cold food.
The rate of diffusion of a gas is inversely proportional to the square root of its density, this is called Graham’s law of diffusion